 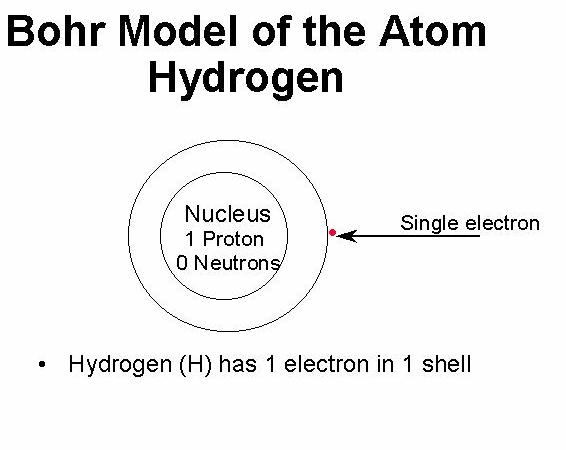 
Electron energies in the hydrogen atom are. This more complex Falstad simulator lets you display the wave functions/orbitals in either real or complex modes, and to take slices along the axes as well as viewing them in three dimensions, potentially valuable for upper level courses analyzing orbital structure. The Bohr model is, however, unable to provide additional details which the full quantum mechanical solution does.Tom Walsh's simulation lets you select individual transitions and view the resulting energy The energy o f the hydrogen atom is the sum of the kinetic and potential energies: E T + V 1 2mv2 e2 r Using Equation 1.7.7, we see that T 1 2V and E 1 2V T This is the form of the virial theorem for a force law varying as r 2.Andrew Duffy’s Emission Spectra simulator lets you compare the hydrogen spectrum with other visible spectra side-by-side with the full white light spectrum, for orientation. 
The PhET model hydrogen atom lets you compare different historical models of the atom to simulated experimental results.We'll use a Bohr diagram to visually represent. Careful analysis shows that, although some of this light is emitted by H 2 molecules, some of the light is also emitted by H atoms. Bohr model of the hydrogen atom attempts to plug in certain gaps as suggested by Rutherfords model by including ideas from the newly developing Quantum. In this video we'll look at the atomic structure and Bohr model for the Hydrogen atom (H). Finally, the hydrogen atom is one of the precious few realistic systemswhich can actually be solved analytically. Bohrs model of the hydrogen atom started from the planetary model, but he added one assumption regarding the electrons. We find that, if we pass a current of electricity through a sample of hydrogen gas, light is emitted. It is a particle in a boxwith spherical, soft walls. Falstad offers this illustration of the energy levels of the hydrogen atoms, which allows you to switch the view between the Bohr and Dirac models within a single active diagram Since hydrogen is the simplest atom, we analyze the hydrogen spectrum first.All the effects that go into fine structure are thus a natural consequence of the Dirac theory. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
